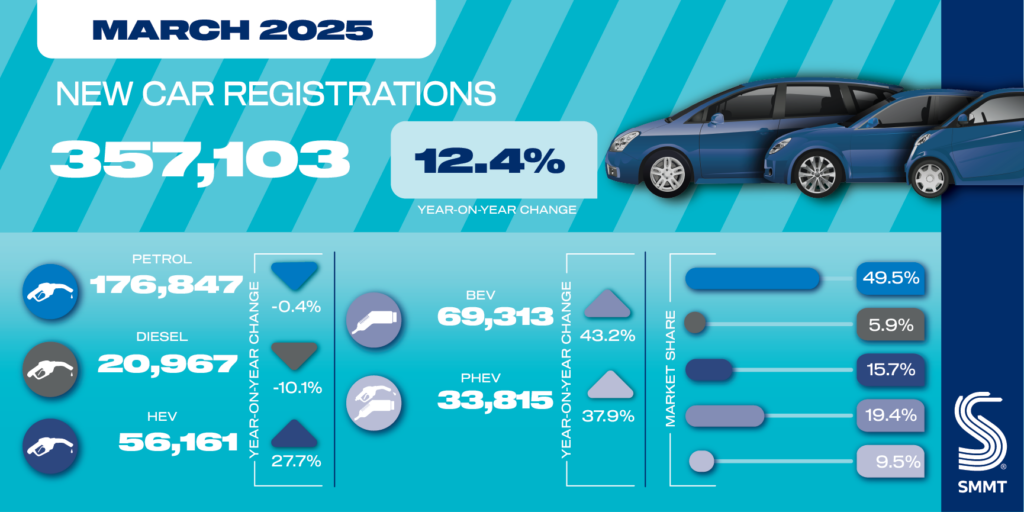
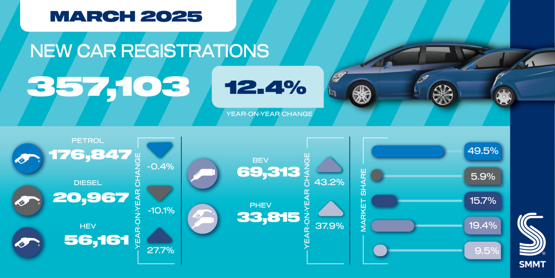
Human medicines European public assessment report (EPAR): Ronapreve, casirivimab,imdevimab, Date of authorisation: 12/11/2021, Revision: 8, Status: Authorised
Human medicines European public assessment report (EPAR): Ronapreve, casirivimab,imdevimab, Date of authorisation: 12/11/2021, Revision: 8, Status: Authorised