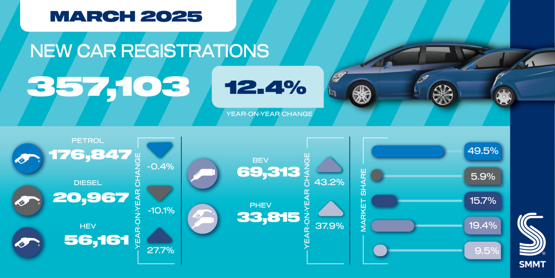
Equillium misses primary endpoint, plus 2 key secondaries, but still plans approval filing
Equillium’s phase 3 graft-versus-host disease trial has missed its primary endpoint and two key secondary objectives. But the biotech brushed off the setback, highlighting other secondary endpoints and post hoc analyses to make the case it could file for FDA approval next year.