This site uses cookies. By continuing to browse the site you are agreeing to our use of cookies.
All
All Stories
All Stories
BioPharma Dive - Latest News
Breaking World Pharma News
Drugs.com - Clinical Trials
Drugs.com - FDA MedWatch Alerts
Drugs.com - New Drug Approvals
Drugs.com - Pharma Industry News
FDA Press Releases RSS Feed
Federal Register: Food and Drug Administration
News and press releases
Pharmaceuticals news FT.com
PharmaTimes World News
Stat
What's new
Preclinical study: after heart attack, a boos...
Mar 30, 2025 0
Enzyme engineering opens door to novel therap...
Mar 30, 2025 0
New tool to boost cancer immunotherapy effects
Mar 27, 2025 0
All
Breaking DefenseFull RSS Feed – Breaking Defense
DefenceTalk
Defense One - All Content
Military Space News
NATO Latest News
The Aviationist
War is Boring
War on the Rocks
NATO Deputy Secretary General calls for stepp...
Apr 3, 2025 0
NATO Secretary General to visit Japan
Apr 3, 2025 0
NATO Foreign Ministers to discuss building a ...
Apr 3, 2025 0
NATO Defence College Field Studies Visit to N...
Apr 2, 2025 0
All
Advanced Energy Materials
CleanTechnica
Energy | FT
Energy | The Guardian
EnergyTrend
Nature Energy
NYT > Energy & Environment
PV-Tech
RSC - Energy Environ. Sci. latest articles
Utility Dive - Latest News
Two‐Step Inverted Perovskite Solar Cells with...
Apr 4, 2025 0
Surface Reconstruction Activates Non‐Noble Me...
Apr 4, 2025 0
A “Cool” Route to Battery Electrode Material ...
Apr 3, 2025 0
Ru Single Atoms Anchored on Co3O4 Nanorods fo...
Apr 3, 2025 0
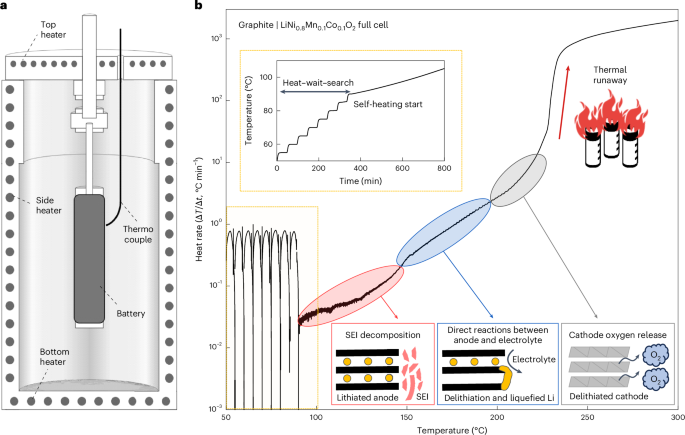
Publisher Correction: Navigating thermal stab...
Mar 28, 2025 0
Rate matters
Mar 25, 2025 0
Amine oxides step up
Mar 25, 2025 0
- Contact
- Agriculture
- Automotive
- Beauty
-
Biopharma
- All
- All Stories
- All Stories
- BioPharma Dive - Latest News
- Breaking World Pharma News
- Drugs.com - Clinical Trials
- Drugs.com - FDA MedWatch Alerts
- Drugs.com - New Drug Approvals
- Drugs.com - Pharma Industry News
- FDA Press Releases RSS Feed
- Federal Register: Food and Drug Administration
- News and press releases
- Pharmaceuticals news FT.com
- PharmaTimes World News
- Stat
- What's new
- Defense
- Energy & Water
- Fashion
- Food & Beverage
- Healthcare
- Legal
- Manufacturing
- Luxury
- Medical Devices
- Mining
- Real Estate
- Retail
- Science Journals
- Transport & Logistics
- Travel & Hospitality