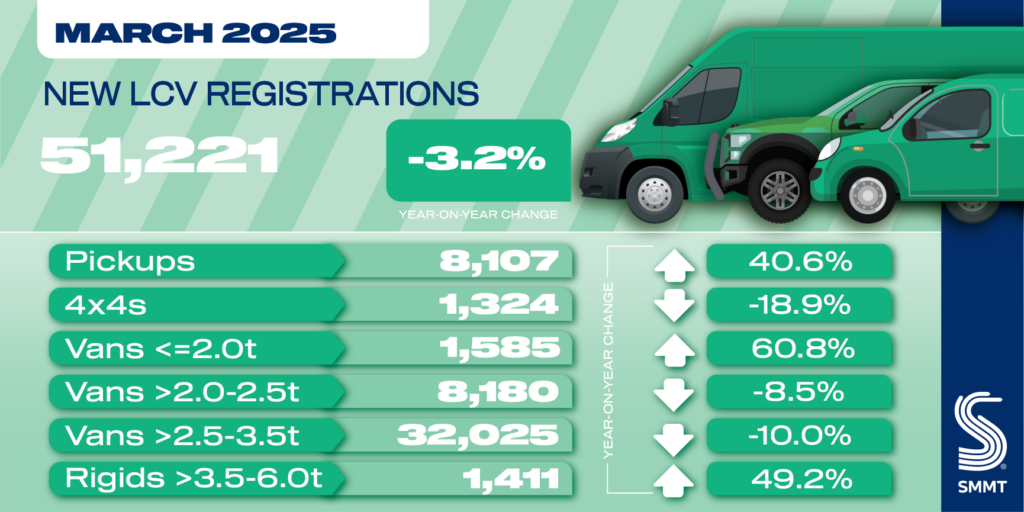
Robust Sodium Storage Enabled by Heterogeneous Engineering and Electrolyte Modification
Advanced Energy Materials, Volume 15, Issue 13, April 1, 2025.

The MoS2/Ti3C2(OH)
x
heterostructure, featuring an enhanced built-in electric field, effectively improves charge transfer and promotes Na+ adsorption. Additionally, the use of 1.0 m NaClO4/1,2-dimethoxyethane electrolyte which exhibits rapid desolvation capabilities contributes to the superior electrochemical performance.
Abstract
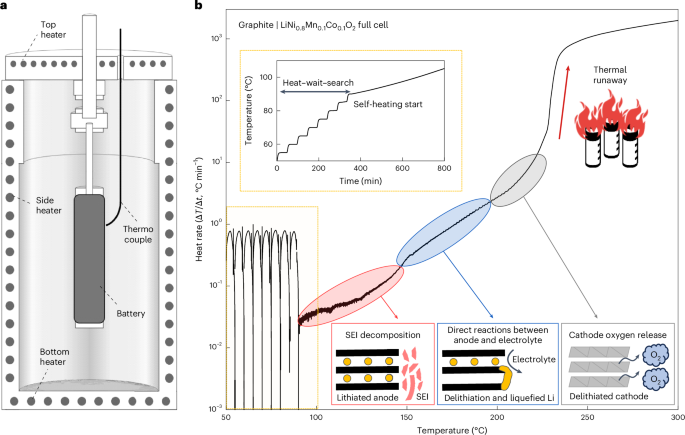
The modulation of heterointerfaces in 2D materials is critically important for improving the electrochemical performance of sodium-ion batteries (SIBs). In this context, the MoS2/Ti3C2T x MXene heterostructure is taken as a typical example to reveal the fundamental principle of high sodium storage performance by regulating the terminal groups of Ti3C2T x . It is demonstrated that MoS2/Ti3C2(OH) x (M/-(OH) x ) heterostructure with a high work function difference generates an enhanced built-in electric field, which facilitates charge transfer. Moreover, ether-based electrolytes, when compared to ester-based electrolytes, provide lower solvation-free energies and exhibit high compatibility with M/-(OH) x , resulting in superior rate capability. Notably, COMSOL simulations of sodium ion (Na+) concentration and Na+ flux distributions reveal that the M/-(OH) x electrode has low concentration polarization and rapid diffusion kinetics in ether-based electrolytes. Consequently, the combination of M/-(OH) x heterostructure with the ether-based electrolyte provides 224.06 mAh g−1 after 1000 cycles at 5.0 A g−1. Furthermore, the Na3V2(PO4)3/C//M/-(OH) x full cell demonstrates robust electrochemical performance, delivering 116.49 mAh g−1 after 140 cycles at 1.0 A g−1. These findings emphasize the impact of modulating terminal functional groups to optimize the electrochemical functionality of heterostructures and highlight the crucial role of electrode/electrolyte synergistic coupling in advancing the practical applications of SIBs.



















































































































































.jpg)



























