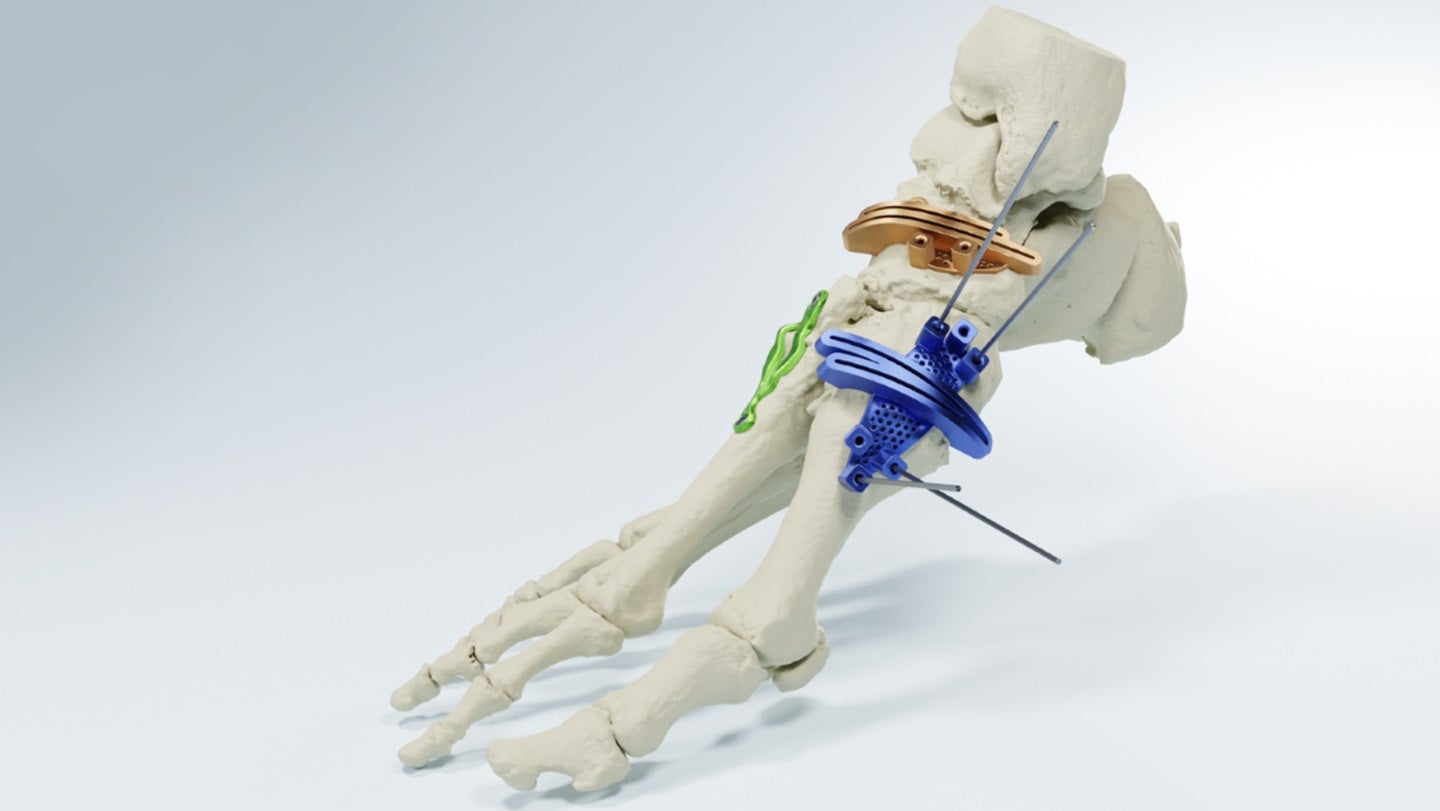
MedCAD secures FDA 510(k) clearance for AccuStride foot and ankle system
MedCAD has received 510(k) clearance from the US Food and Drug Administration (FDA) for its AccuStride Foot and Ankle System. The post MedCAD secures FDA 510(k) clearance for AccuStride foot and ankle system appeared first on Medical Device Network.
The post MedCAD secures FDA 510(k) clearance for AccuStride foot and ankle system appeared first on Medical Device Network.








































































































































































.jpg)