Tuning Polysulfides into Clustered‐States via Non‐coordinating Molecular Encapsulation to Achieve an Alternative Kinetics in Li─S Batteries
Advanced Energy Materials, Volume 15, Issue 23, June 17, 2025.

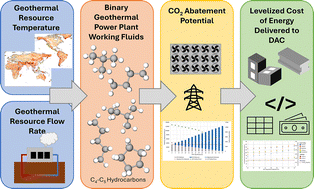
Herein, a clustered polysulfides-encapsulated (CPE) mechanism is proposed, in which the polysulfides are allowed to form clusters by encapsulation with uncoordinated aromatic solvents. The clustered-polysulfide encapsulation (CPE) species where the TFSI− anions are mediated in the CPE species become the new basic reactive units. The CPE mechanism enables alternative reaction kinetics and incorporates the deposition process of Li2S.
Abstract
The practical application of lithium–sulfur batteries is hindered by the polysulfide shuttle effect and sluggish kinetics inherent in solid–liquid–solid conversion mechanism, particularly under lean electrolyte conditions (<5 µL mg⁻¹). Weakly solvating electrolytes and localized high-concentration electrolytes can suppress polysulfide dissolution and enable a quasi-solid-phase mechanism but suffer from severely limited reaction kinetics. Herein, a clustered-polysulfide-mediated sulfur conversion mechanism enabled by a novel electrolyte composed of 1,2-dimethylbenzene (DTL) and 1,2-dimethoxyethane (DME) is proposed. The encapsulation effect of DTL and the coordination of TFSI⁻ with polysulfides drives the aggregation of polysulfides so that the clustered polysulfides with virtual shell boundaries can be the new basic reactive that bridges the gap between the traditional dissolution-dominated mechanism and quasi-solid-state mechanism. The clustered polysulfide electrolyte (CPE) not only suppresses the shuttle effect and stabilizes the lithium anode by mitigating parasitic reactions but also enables alternative reaction kinetics and promotes 3D Li₂S deposition, minimizing electrode passivation. Ultimately, lithium–sulfur batteries can achieve excellent electrochemical performance and can stably operate under lean electrolyte (<4.0 µL mg⁻¹) with an area capacity of >4 mAh cm−2. This work elucidates the relationship between polysulfide dissolution behavior and redox kinetics, providing a new insight into the understanding of complex sulfur conversion mechanisms.














































































![The sights of Paris Air Show, one last time: Day 4 [Photos]](https://breakingdefense.com/wp-content/uploads/sites/3/2025/06/20250617-helenedelacoste-Paris-Air-Show-037-scaled-e1750357690820.jpg?#)