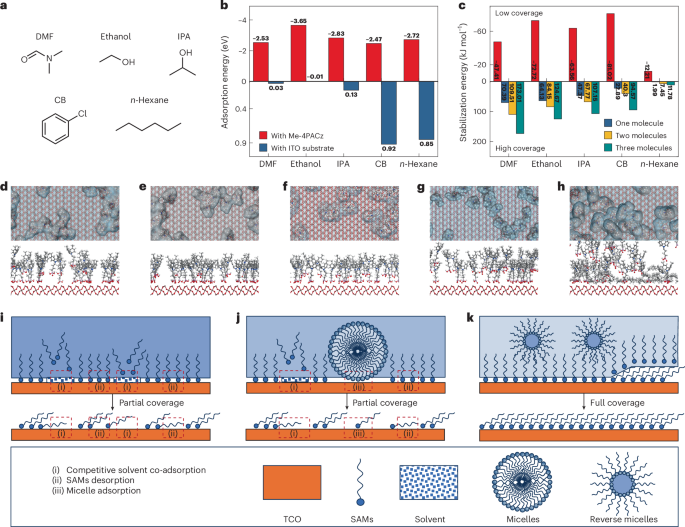
Interaction of cardiac leiomodin with the native cardiac thin filament
by Madison Little, Cristina M. Risi, Tania M. Larrinaga, Mason Summers, Tyler Nguyen, Garry E. Smith Jr, Jennifer Atherton, Carol C. Gregorio, Alla S. Kostyukova, Vitold E. Galkin Every heartbeat depends on cyclical contraction-relaxation produced by the interactions between myosin-containing thick and actin-based thin filaments (TFs) arranged into a crystalline-like lattice in the cardiac sarcomere. Therefore, the maintenance of thin filament length is crucial for myocardium function. The thin filament is comprised of an actin backbone, the regulatory troponin complex and tropomyosin that controls interactions between thick and thin filaments. Thin filament length is controlled by the tropomodulin family of proteins; tropomodulin caps pointed ends of thin filaments, and leiomodin (Lmod) promotes elongation of thin filaments by a “leaky-cap” mechanism. The broader distribution of Lmod on the thin filament implied to the possibility of its interaction with the sides of thin filaments. Here, we use biochemical and structural approaches to show that cardiac Lmod (Lmod2) binds to a specific region on the native cardiac thin filament in a Ca2+-dependent manner. We demonstrate that Lmod2’s unique C-terminal extension is required for binding to the thin filament actin backbone and suggest that interactions with the troponin complex assist Lmod2’s localization on the surface of thin filaments. We propose that Lmod2 regulates the length of cardiac thin filaments in a working myocardium by protecting newly formed thin filament units during systole and promoting actin polymerization at thin filament pointed ends during diastole.
by Madison Little, Cristina M. Risi, Tania M. Larrinaga, Mason Summers, Tyler Nguyen, Garry E. Smith Jr, Jennifer Atherton, Carol C. Gregorio, Alla S. Kostyukova, Vitold E. Galkin Every heartbeat depends on cyclical contraction-relaxation produced by the interactions between myosin-containing thick and actin-based thin filaments (TFs) arranged into a crystalline-like lattice in the cardiac sarcomere. Therefore, the maintenance of thin filament length is crucial for myocardium function. The thin filament is comprised of an actin backbone, the regulatory troponin complex and tropomyosin that controls interactions between thick and thin filaments. Thin filament length is controlled by the tropomodulin family of proteins; tropomodulin caps pointed ends of thin filaments, and leiomodin (Lmod) promotes elongation of thin filaments by a “leaky-cap” mechanism. The broader distribution of Lmod on the thin filament implied to the possibility of its interaction with the sides of thin filaments. Here, we use biochemical and structural approaches to show that cardiac Lmod (Lmod2) binds to a specific region on the native cardiac thin filament in a Ca2+-dependent manner. We demonstrate that Lmod2’s unique C-terminal extension is required for binding to the thin filament actin backbone and suggest that interactions with the troponin complex assist Lmod2’s localization on the surface of thin filaments. We propose that Lmod2 regulates the length of cardiac thin filaments in a working myocardium by protecting newly formed thin filament units during systole and promoting actin polymerization at thin filament pointed ends during diastole.