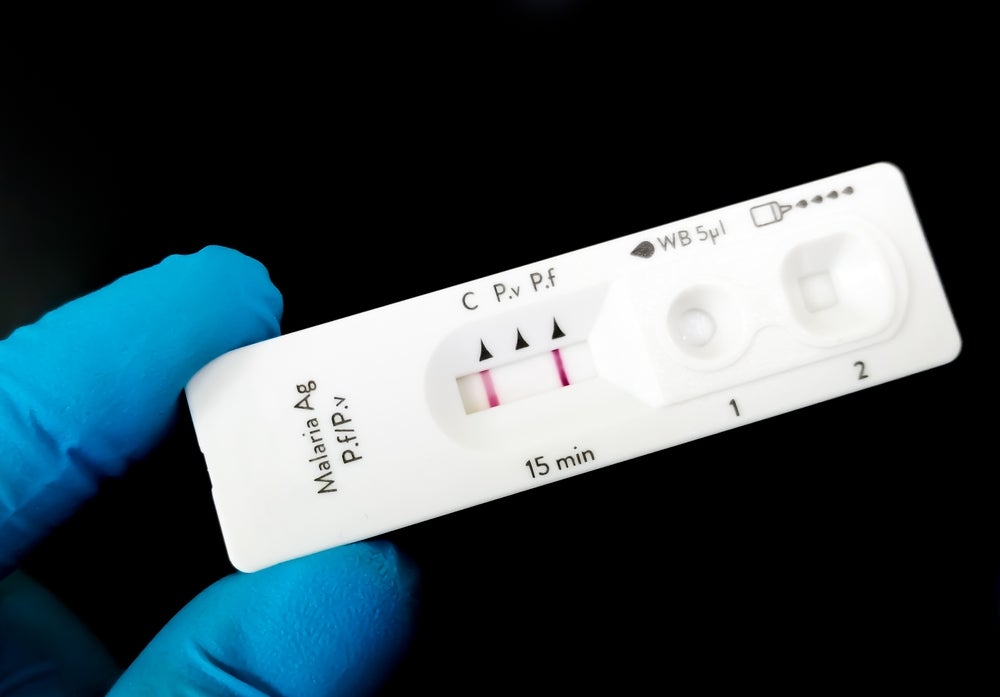
FDA grants expanded 510(k) clearance for Epitel’s wireless EEG system
The US Food and Drug Administration (FDA) has granted expanded 510(k) clearance for Epitel’s REMI Wireless electroencephalogram (EEG) System. The post FDA grants expanded 510(k) clearance for Epitel’s wireless EEG system appeared first on Medical Device Network.

The post FDA grants expanded 510(k) clearance for Epitel’s wireless EEG system appeared first on Medical Device Network.








































































































































































.jpg)