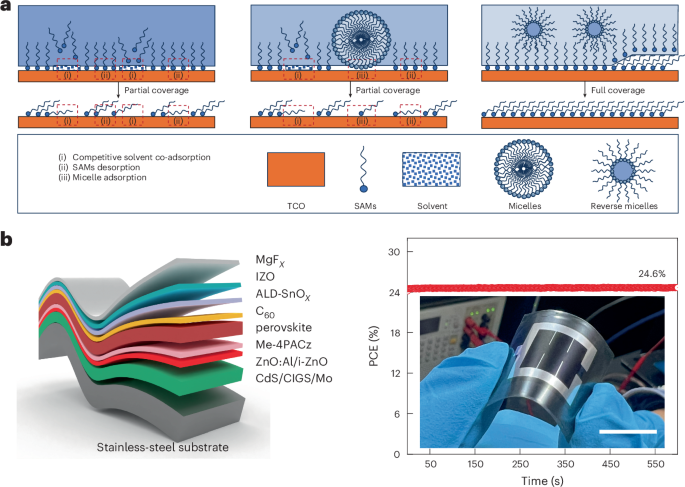
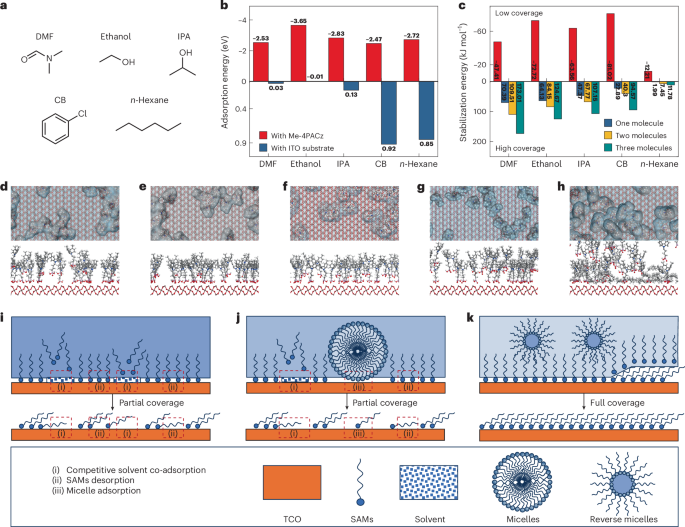
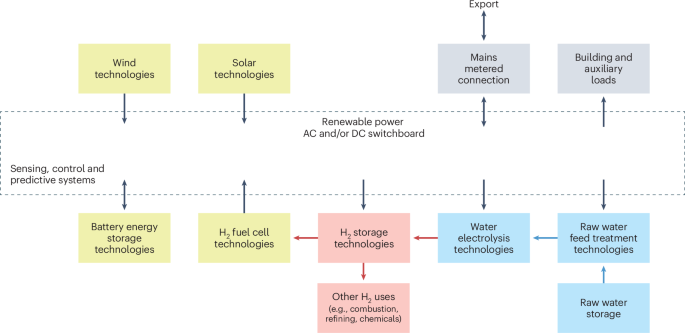
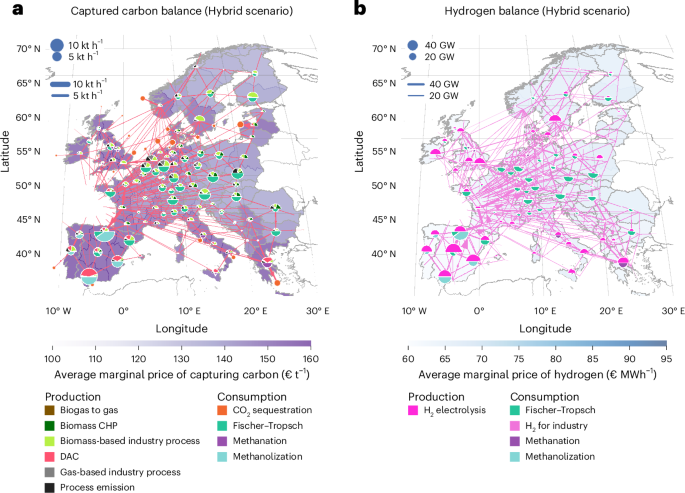
FDA Approves Stoboclo (denosumab-bmwo), a Biosimilar to Prolia
JERSEY CITY, N.J., March 3, 2025 /PRNewswire/ -- Celltrion today announced that the U.S. Food and Drug Administration (FDA) has approved Stoboclo (CT-P41, denosumab-bmwo), a biosimilar referencing Prolia (denosumab), for all indications of the...