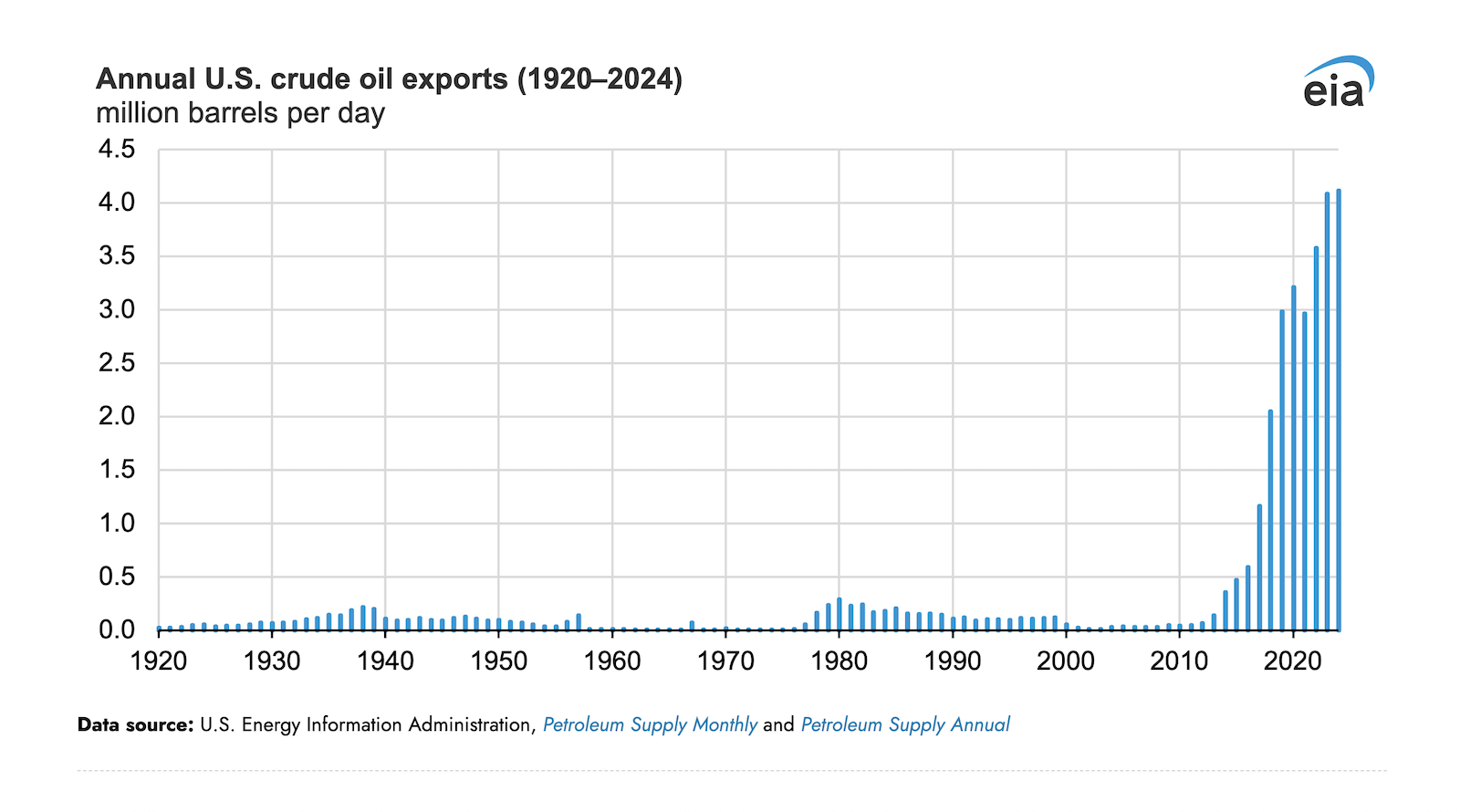
FDA Approves Opdivo Qvantig (nivolumab and hyaluronidase-nvhy) Subcutaneous Injection for Use in Most Previously Approved Solid Tumor Opdivo (nivolumab) Indications
PRINCETON, N.J.--(BUSINESS WIRE) December 27, 2024 -- Bristol Myers Squibb (NYSE: BMY) today announced that the U.S. Food and Drug Administration (FDA) granted approval for Opdivo Qvantig (nivolumab and hyaluronidase-nvhy) injection for...