Spatial‐Constraint Modulation of Intra/Extracellular Reactive Oxygen Species by Adaptive Hybrid Materials for Boosting Pyroptosis and Combined Immunotherapy of Breast Tumor
Advanced Healthcare Materials, EarlyView.

A synthesized hybrid material system addresses ROS requirements in dynamic sono/immuno-therapy processes. Within cells, the activated nanoparticles trigger Ca2+ release and ROS generation under US, leading to pyroptosis and a robust immune response. The hydrogel retained in the extracellular matrix is employed to efficiently control the secondary ROS, thereby prolonging the activity of cytotoxic T cells.
Abstract
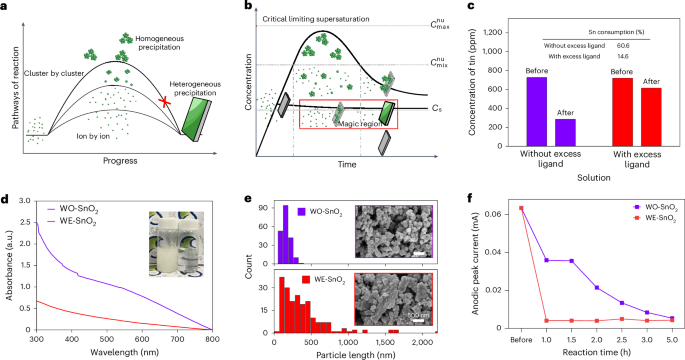
Pyroptosis-immunotherapy has potential for triple-negative breast cancer treatment, but its efficacy is limited by insufficient pyroptosis activation and the need for phased, balanced, and spatially controlled activation of active species during long-term treatment. To reconcile intracellular/extracellular demands in tumor ablation, a nanoparticle-hydrogel hybrid enabling spatiotemporal reactive oxygen species (ROS) modulation is engineered. An open-shell sonosensitizer with unpaired electrons in its molecular orbitals is prepared by chelating Cu2⁺ with TCPP. These sonosensitizers are undergoing bovine serum albumin mediated biomineralization to form calcium phosphate particles and are incorporated into an injectable hydrogel through Schiff base crosslinking between dopamine-functionalized oxidized hyaluronic acid and gallic acid-modified chitosan. After intratumoral injection, nanoparticles endocytosed into tumor cells undergo acidic degradation, releasing calcium ions and GSH-activatable sonosensitizers. Calcium overload synergizes with ultrasound-mediated oxidative stress to induce mitochondrial damage and pyroptosis, while adhesive hydrogels retained in the extracellular matrix control excessive secondary ROS levels to protect oxidation-sensitive entities. This dual-action mechanism enhances the overall therapeutic effect by combining immediate tumor killing with long-term immune activation. This study provides a new route to hybrid material design, addressing the conflicting demands of short-term tumor ablation and long-term immune activation, overcoming the limitations of current pyroptosis-based immunotherapies.